Covalent Bonds definition
Another method of forming bonds between atoms is electron sharing. The atoms acquire the noble gas configuration by sharing electrons in this manner. A covalent bond is the joining of atoms by sharing electrons between two atoms.
Formation of homoatomic molecules and heteroatomic molecules
Homoatomic molecules
The sharing of electrons between atoms of the same type produces homoatomic molecules.
Examples for homoatomic molecules
- Oxygen (O2)
- Hydrogen (H2)
- Fluorine (F2)
- Nitrogen (N2)
Heteroatomic molecules
The sharing of electrons between atoms of different elements results in the formation of heteroatomic molecules.
Examples for heteroatomic molecules
methane (CH4), water (H2O), ammonia (NH3)
Examples of compounds that have covalent bonds
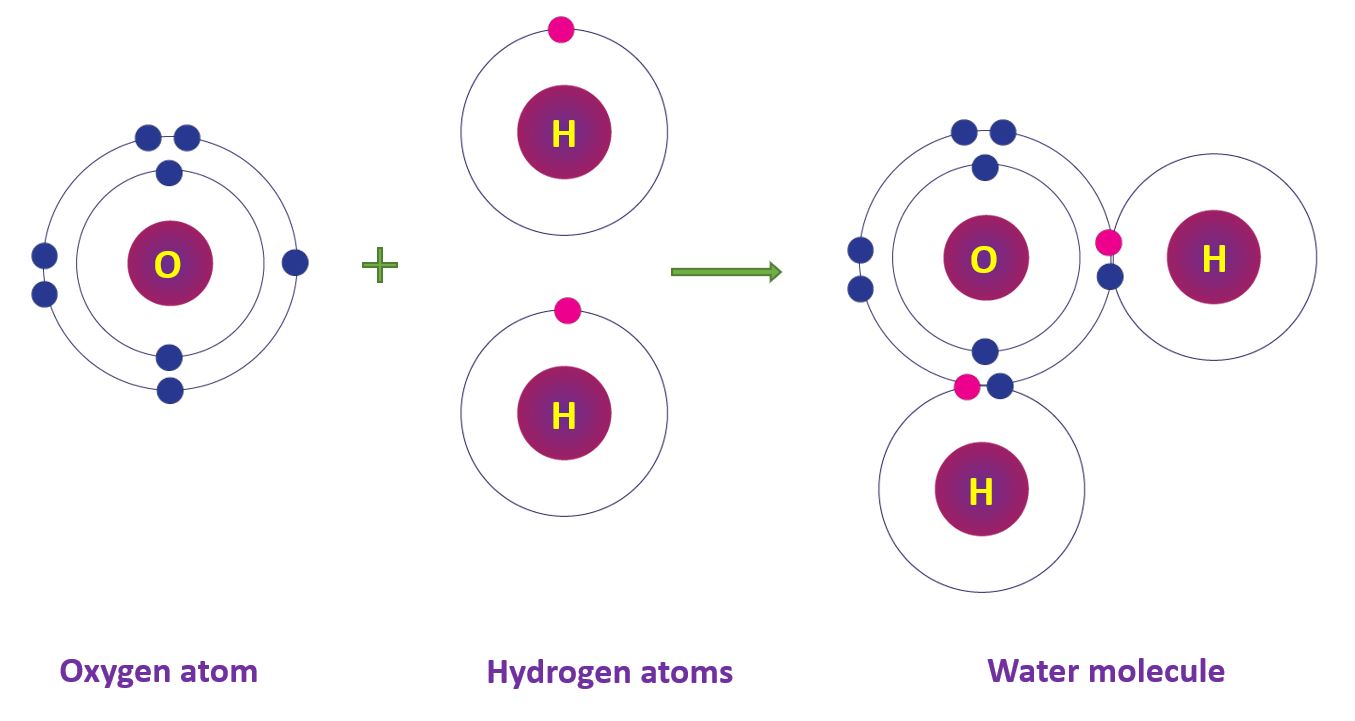
Covalent Bonds in Water molecule
The oxygen atom’s electronic configuration is 2, 6. An oxygen atom shares two pairs of electrons with two hydrogen atoms, resulting in the formation of two single bonds and the formation of the water (H2O) molecule.
Covalent Bonds in Water molecule in a diagram
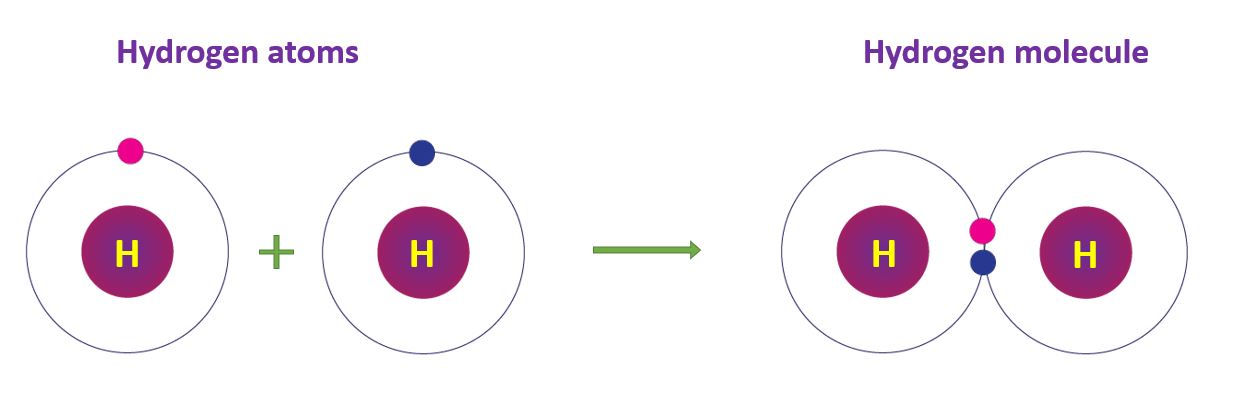
Covalent Bonds in Hydrogen molecule
One electron exists in a hydrogen atom. Two hydrogen atoms share their electrons, resulting in the stable configuration of helium. This results in the hydrogen molecule (H2), which is formed when two hydrogen atoms form a covalent bond.
Covalent Bonds in Hydrogen molecule in a diagram
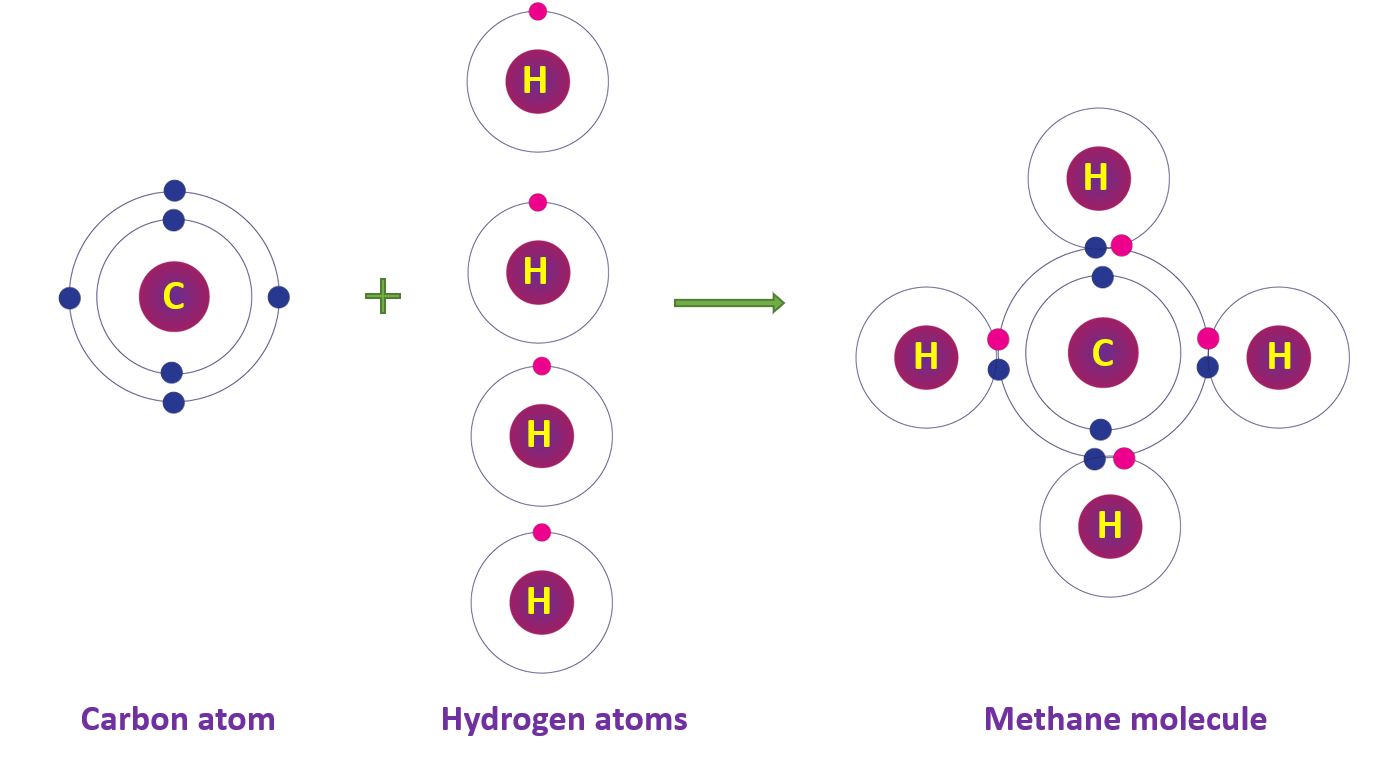
Covalent Bonds in Methane molecule
The carbon atom’s electronic configuration is 2, 4. Four hydrogen atoms share four electrons with a carbon atom to form a four-single-bond methane molecule (CH4).
Covalent Bonds in Methane molecule in a diagram
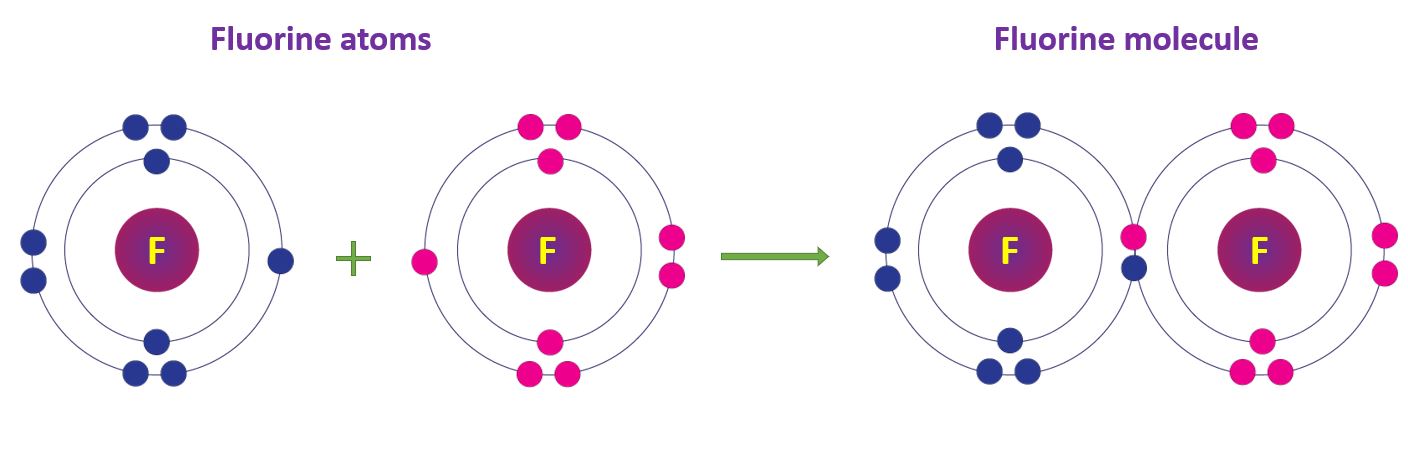
Covalent Bonds in Fluorine molecule
A fluorine atom has an electronic configuration of 2, 7. Two fluorine atoms achieve the stable electronic configuration by sharing a pair of electrons. As a result, two fluorine atoms are covalently bonded to form a fluorine molecule.
Covalent Bonds in Fluorine molecule in a diagram
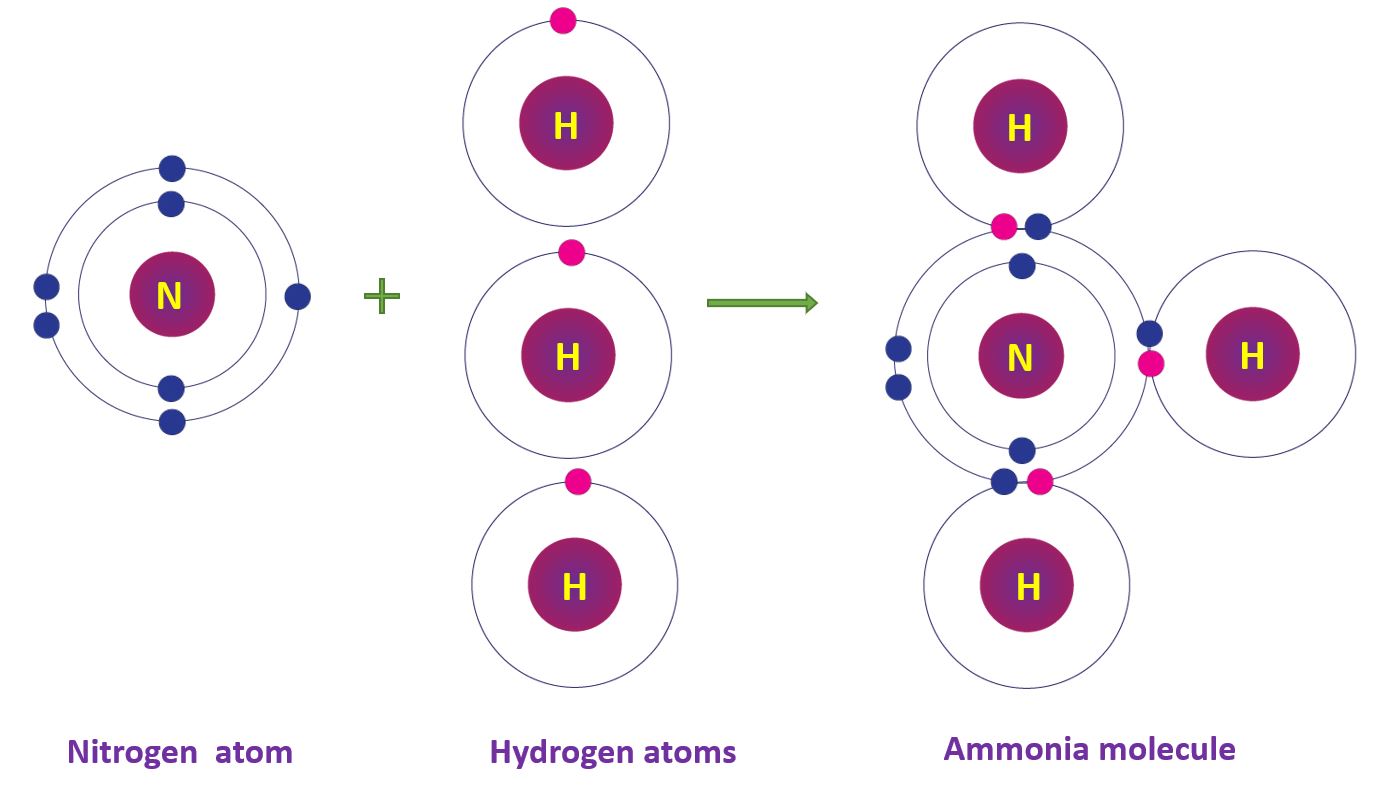
Covalent Bonds in Ammonia molecule
The nitrogen atom has an electronic configuration of 2, 5. Three hydrogen atoms share three electron pairs with one nitrogen atom. As a result, an ammonia (NH3) molecule with three single bonds is formed.
Covalent Bonds in Ammonia molecule in a diagram
Covalent Bonds in Carbon tetrachloride molecule
A carbon atom’s electronic configuration is 2, 4. A chlorine atom’s electronic configuration is 2, 7. A carbon atom and four chlorine atoms share four pairs of electrons to form the carbon tetrachloride (CCl4) molecule.
Covalent Bonds in Hydrogen chloride molecule
A chlorine atom’s electronic configuration is 2, 8, 7. A chlorine atom and a hydrogen atom share a pair of electrons to form the hydrogen chloride (HCl) molecule.
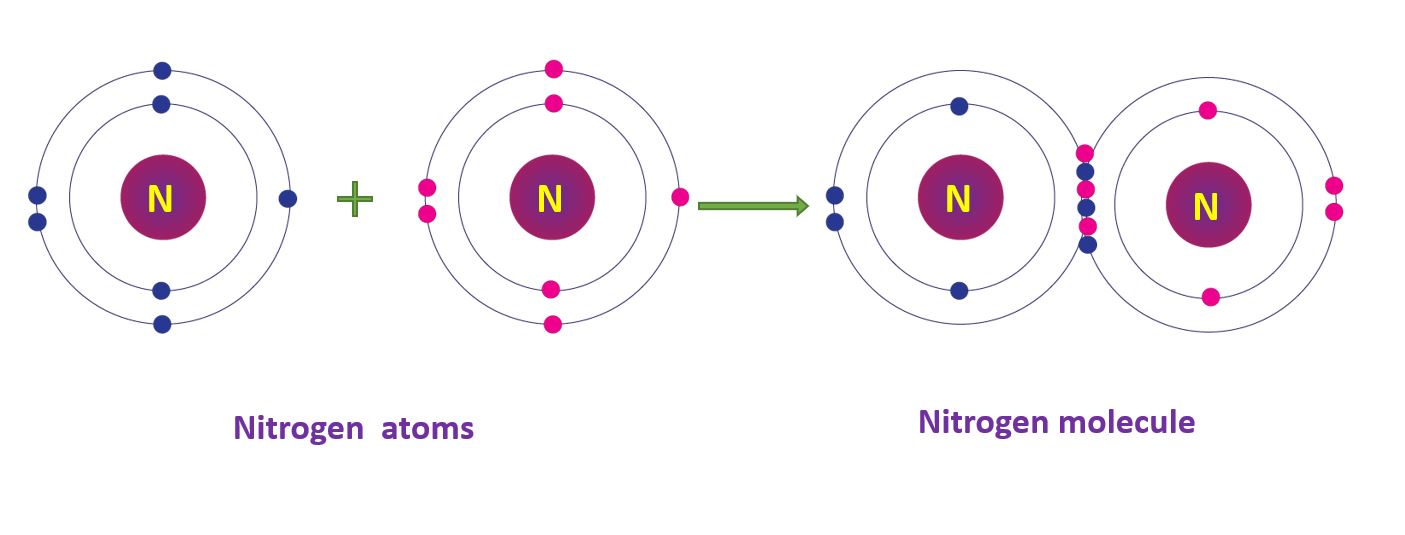
Covalent Bonds in Nitrogen molecule
A nitrogen atom’s electronic configuration is 2, 5. By sharing three pairs of electrons, two nitrogen atoms form a nitrogen (N2) molecule. A triple bond is so named because it shares three pairs of electrons.
Covalent Bonds in Nitrogen molecule in a diagram
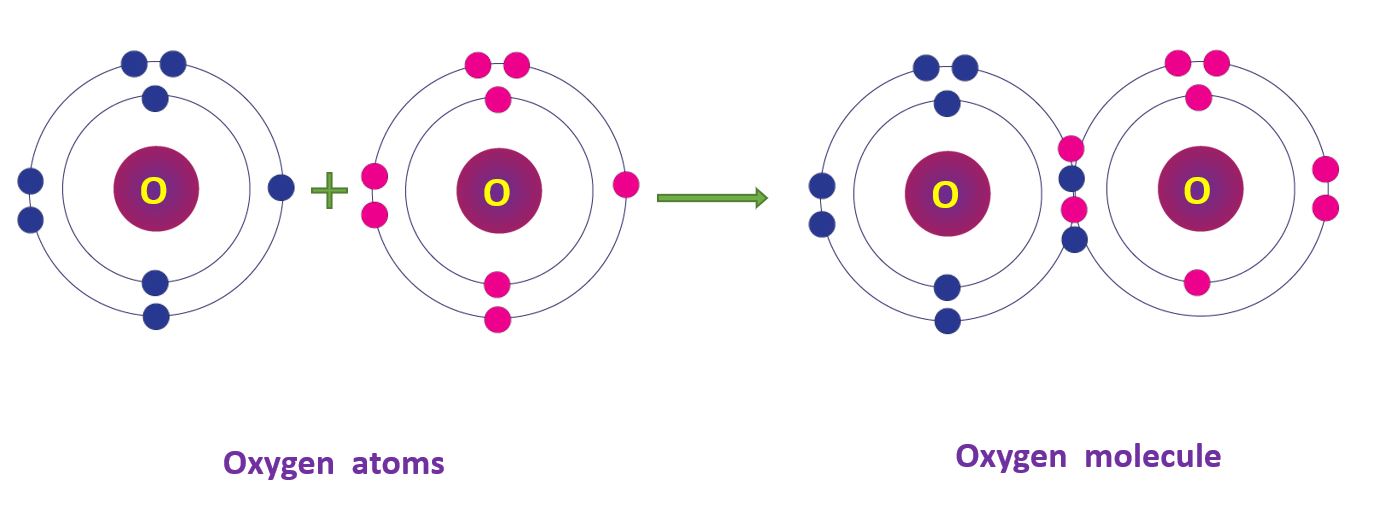
Covalent Bonds in Oxygen molecule
The electronic configuration of the oxygen atom – 2, 6. Two oxygen atoms share two pairs of electrons when forming the oxygen (O2) molecule. Because two pairs of electrons are shared, the bond is referred to as a double bond.
Covalent Bonds in Oxygen molecule in a diagram
Atomic Lattice
Atoms in some elements are organized as a lattice. Atomic lattices are lattices in which the atoms are covalently bonded. Carbon occurs naturally in two atomic lattices: graphite and diamond. They are known as allotropic carbon forms. The way the carbon atoms form covalent bonds with one another differs between these two forms. Covalent compounds have low melting and boiling points in general. Diamond and graphite, on the other hand, have high melting and boiling points due to their atomic lattice structure.
Forms of carbon atomic lattice
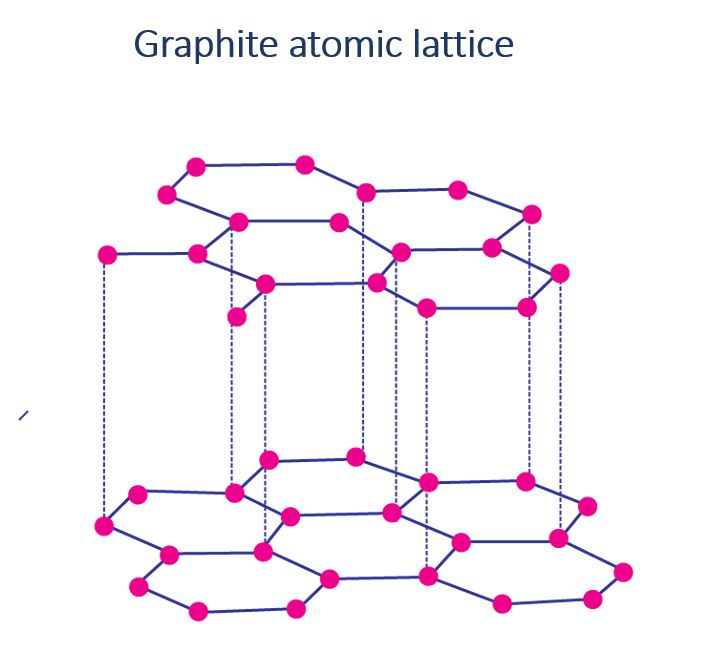
Graphite atomic lattice
Graphite is made up of layers of carbon atoms formed by single bonds connecting one carbon atom to three other carbon atoms. These layers are layered on top of one another. These layers are held together by weak forces. As a result, one layer easily slides over the other. Graphite acts as a lubricant due to its structure.
Graphite atomic lattice in a diagram
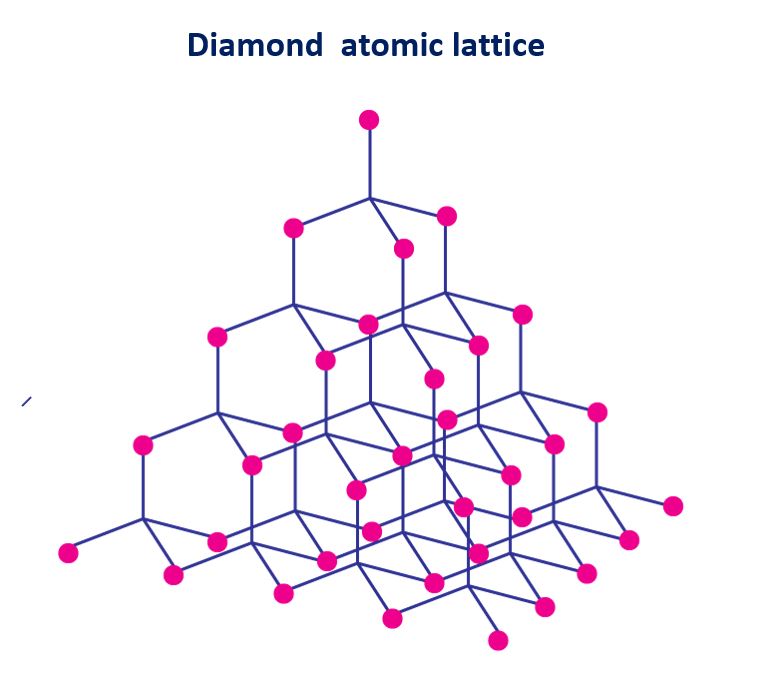
Diamond atomic lattice
Every carbon atom in diamond forms four single bonds with four other carbon atoms in a three-dimensional lattice. The hardest substance found in nature is diamond.
Diamond atomic lattice in a diagram
Polar vs nonpolar covalent bonds
Covalent bonds are chemical bonds formed by the sharing of electrons between two atoms. Depending on the electronegativity difference between the two atoms in a covalent bond, the bond can be polar or nonpolar.
In a nonpolar covalent bond, the electrons are shared equally between the two atoms, and there is no separation of charge. This occurs when the two atoms have similar electronegativities, such as in the bond between two atoms of the same element, such as H2 or O2.
Examples of polar covalent bonds
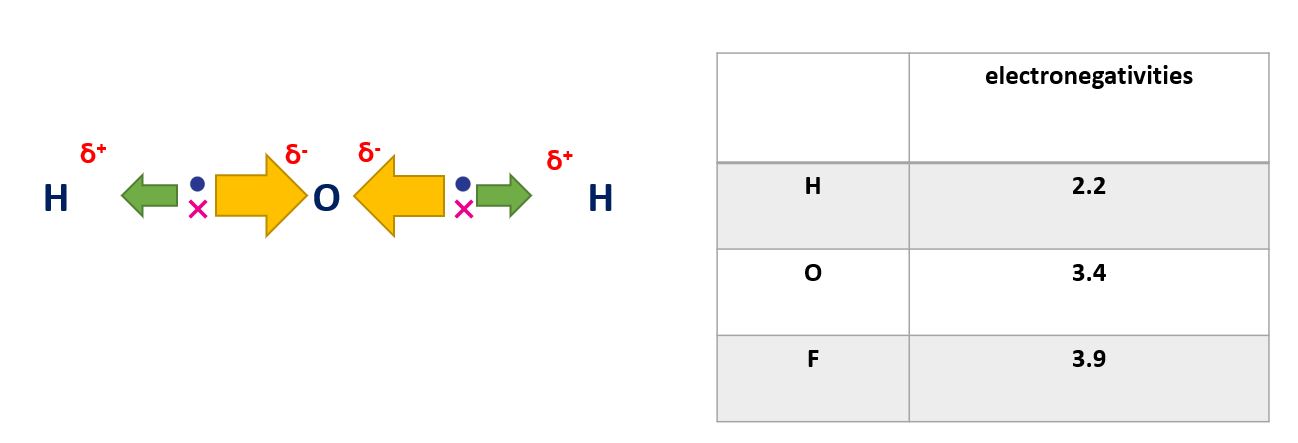
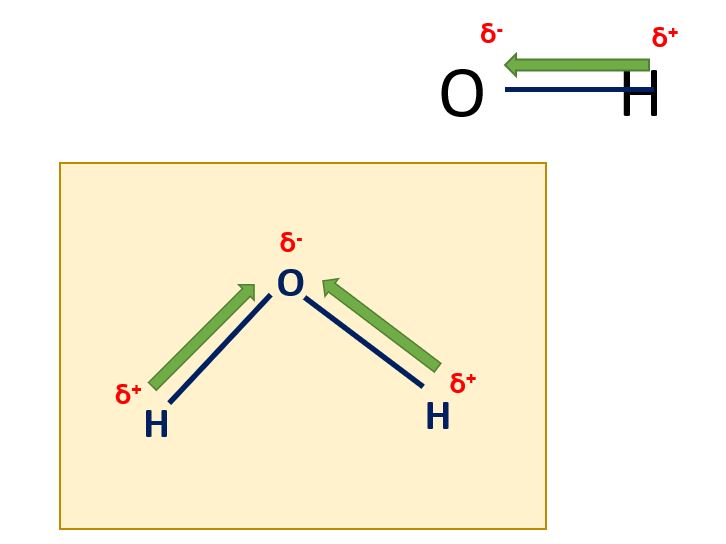
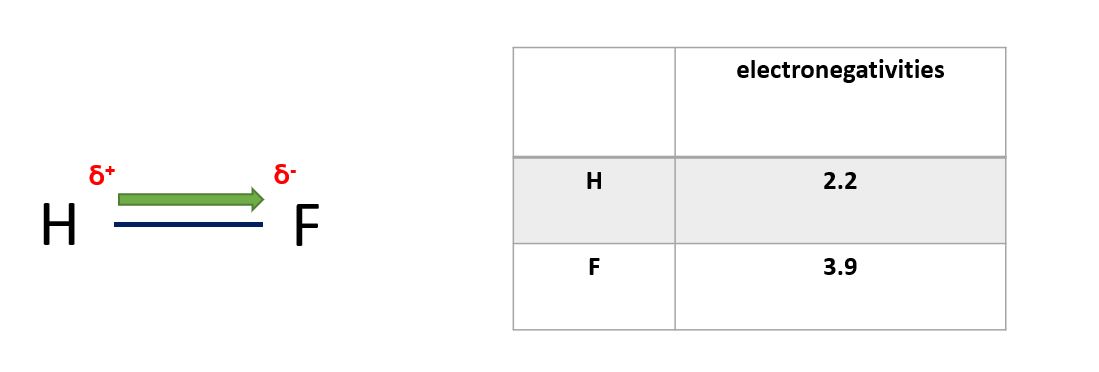
In a polar covalent bond, the electrons are shared unequally between the two atoms, resulting in a separation of charge. This occurs when the two atoms have different electronegativities, such as in the bond between hydrogen and oxygen in water (H2O). The oxygen atom has a higher electronegativity and therefore attracts the shared electrons more strongly, creating a partial negative charge on the oxygen atom and a partial positive charge on the hydrogen atoms.
Polar covalent bonds in a diagram
Polar covalent bonds of water in a diagram
Polar covalent bonds in a diagram
Properties of polar covalent bonds
The polarity of a covalent bond has significant ramifications for the molecule’s characteristics. Polar molecules, for example, have higher boiling temperatures than nonpolar molecules because they can form stronger intermolecular interactions. Polar molecules can dissolve in polar solvents as well, but nonpolar molecules can only dissolve in nonpolar solvents.
How many covalent bonds can carbon form
Carbon can form up to four covalent bonds. This is because carbon has four valence electrons in its outermost shell, which are available for bonding. By sharing these electrons with other atoms, carbon can form stable covalent compounds. In organic chemistry, carbon is the backbone of many important molecules, such as hydrocarbons, carbohydrates, proteins, and nucleic acids. The ability of carbon to form multiple covalent bonds is a key factor in the diversity and complexity of organic molecules.